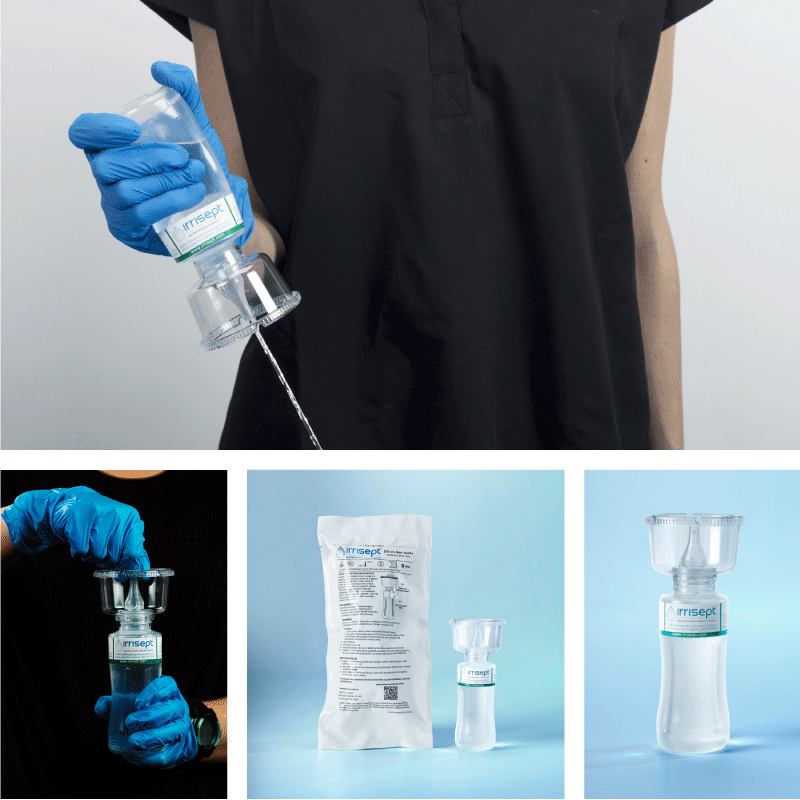
Irrisept Antimicrobial Wound Lavage is available to order for irrigation needs to address the national saline shortage.
Please visit our ORDER page for next steps.
Products
View current products and learn more about Irrisept®
A New Standard of Care for Wound Irrigation
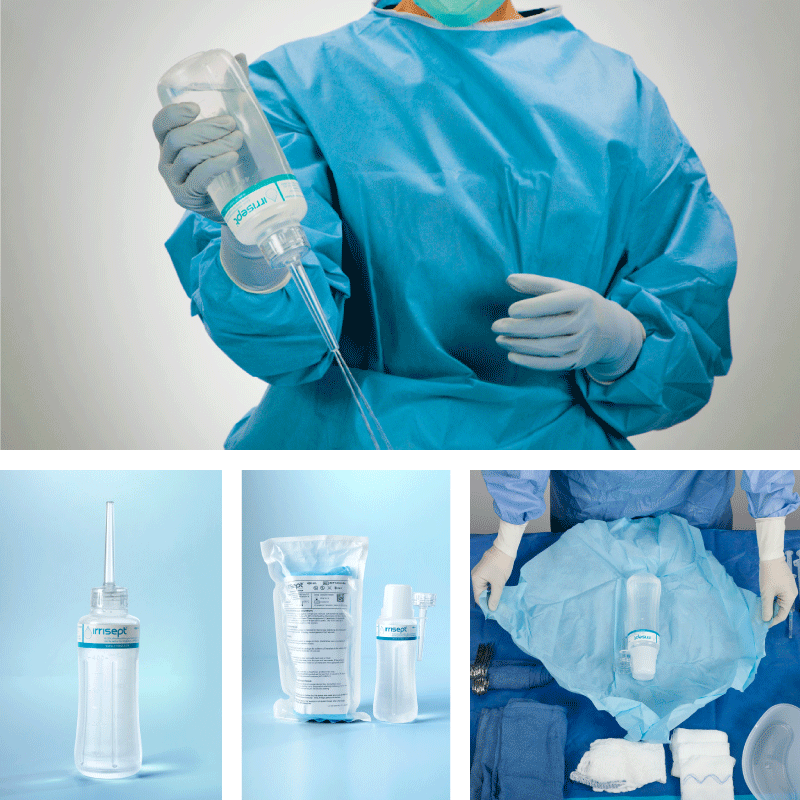
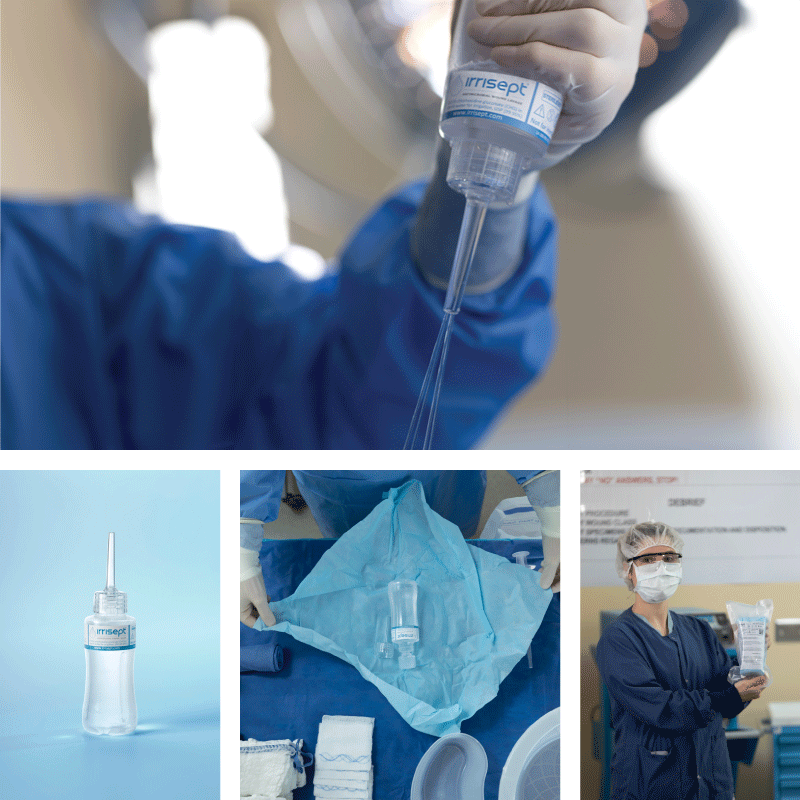
Irrisept is a self-contained jet lavage that contains chlorhexidine gluconate (CHG) as a preservative to offer broad-spectrum activity against various microorganisms in the bottled solution.1
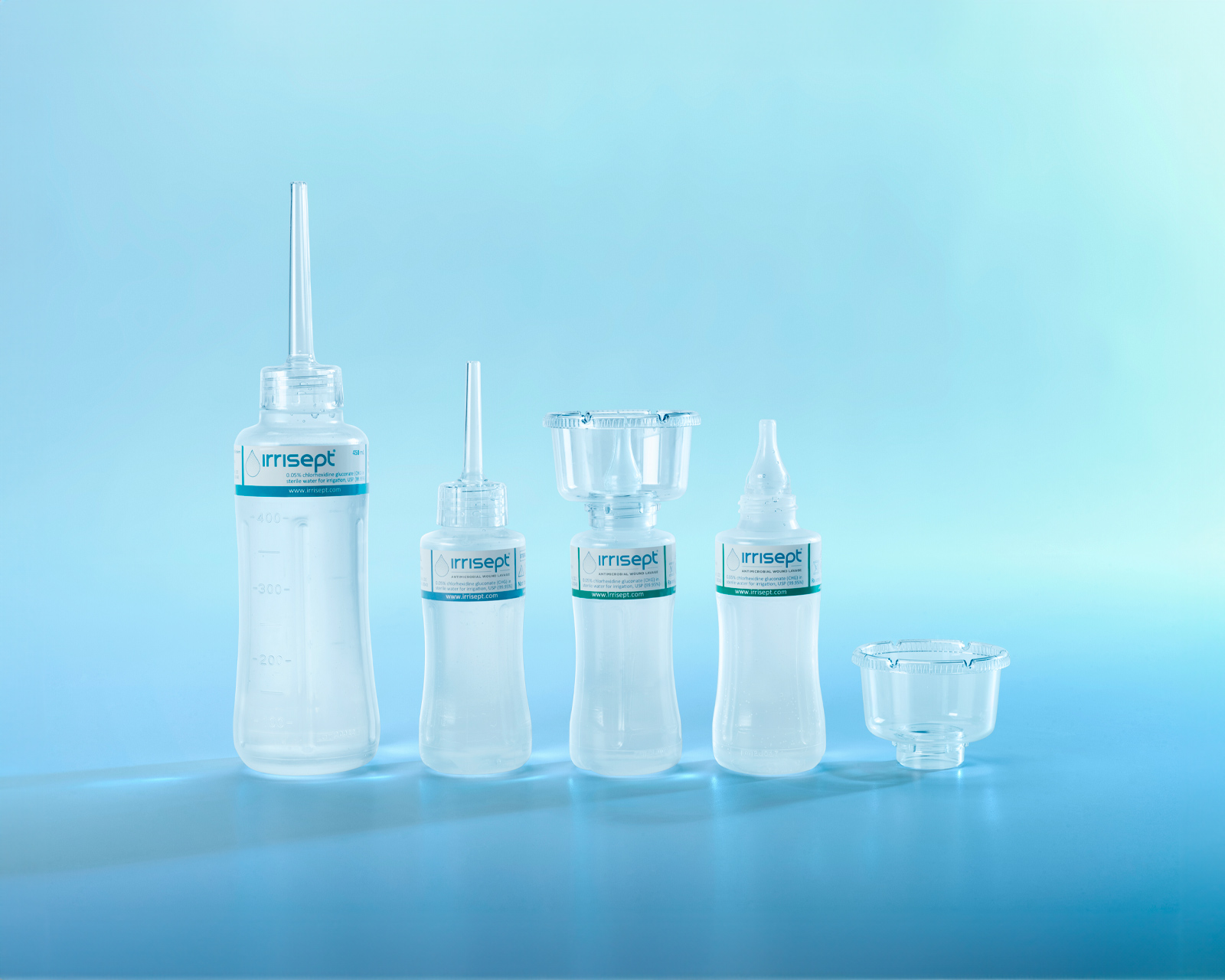
Product Sizes/Packaging
Irrisept Antimicrobial Wound Lavage is offered in a variety of sizes and configurations to meet your clinical needs.
Irrisept is intended for mechanical cleansing and removal of debris, dirt, and foreign materials, including microorganisms from wounds.
Warning: Do not use this product if the patient is allergic to chlorhexidine gluconate. Discontinue use immediately if irritation, sensitization, or allergic reaction occurs.
Sterile Packaged
Irrisept Antimicrobial Wound Lavage
450 mL, Sterile Packaged
- Manufacturer #: ISEPT-450-USA
- Includes: (1) 450mL bottle of Irrisept, (1) Irriprobe applicator, and (1) Convenience cap in sterile packaging
- 12 bottles per case
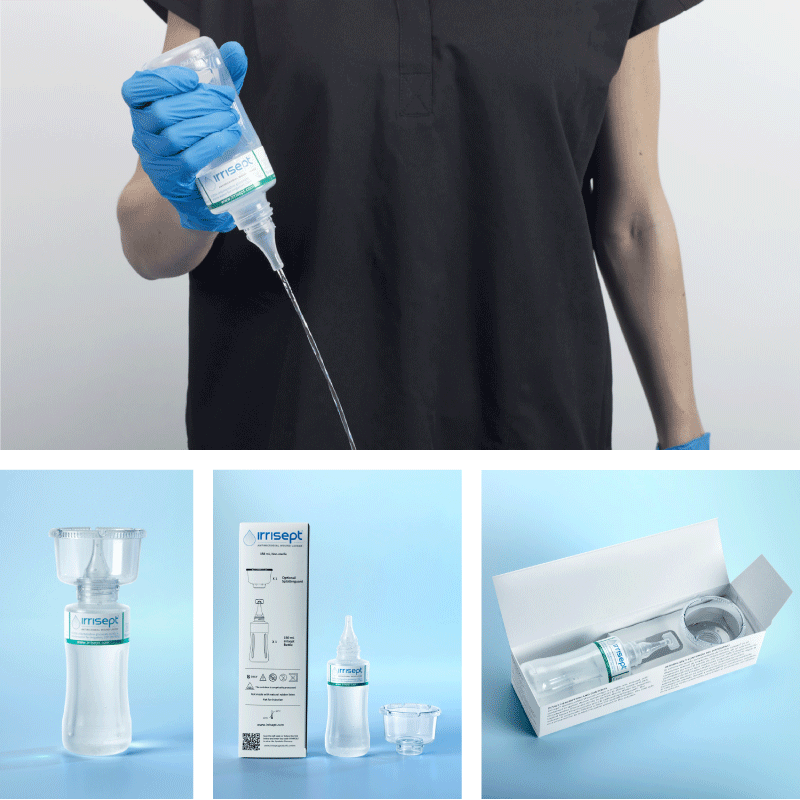
Irrisept Antimicrobial Wound Lavage
150 mL, Sterile Packaged
- Manufacturer #: ISEPT-150-USA
- Includes: (1) 150mL bottle of Irrisept and (1) Irriprobe applicator in sterile packaging.
- 15 bottles per case
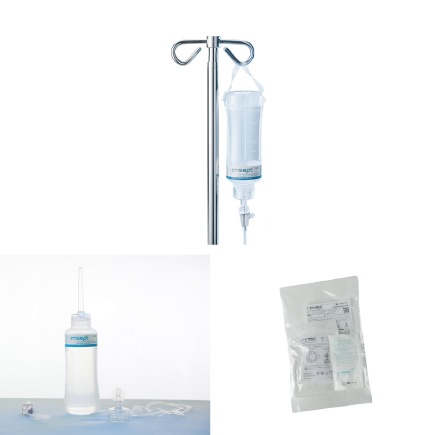
Irrisept Accessory Kit
Spikeable connection, Sterile Packaged (to be connected to ISEPT-450-USA)
- Manufacturer #: ISEPT-AK-USA
- Includes: (1) Vented Spike, (1) Spikable Cap, (2) Bottle Hangers
- 24 kits per case
Advanced Wound Care
Irrisept Antimicrobial Wound Lavage
150 mL, Post Acute Wound Care
- Manufacturer #: ISEPT-150N-USA
- Includes: (1) 150mL bottle of Irrisept with (1) attachable Splatterguard in an easy-to-store shelf carton
- 15 bottles per case
Irrisept Antimicrobial Wound Lavage
150 mL, Tear & Go Pouch
- Manufacturer #: ISEPT-150RP-USA
- Includes: (1) 150mL bottle of Irrisept with (1) detachable Splatterguard in a Tear & Go Pouch
- 12 bottles per case
Irrisept Wound Solution Kit
Instillable configuration of 450mL bottle of Irrisept Antimicrobial Wound Lavage
- Manufacturer #: ISEPT-WS-USA
- Includes: (1) 450mL bottle of Irrisept, (1) Spikable Cap, (2) Bottle Hangers
- 10 kits per case
FAQ
What is Irrisept®?
Irrisept Antimicrobial Wound Lavage is a single-use, self-contained irrigation device. Irrisept contains 0.05% chlorhexidine gluconate (CHG) in 99.95% Sterile Water for Irrigation, United States Pharmacopeia (USP). The solution is aseptically filled in a Blow-Fill-Seal (BFS) bottle. The CHG acts as a preservative to inhibit microbial growth in the bottled solution. Irrisept can be delivered through manual compression or spikeable/instillable connections with powered irrigation devices. Irrisept is a Class II Medical Device and an unclassified Combination Product.
What is CHG?
CHG is chlorhexidine gluconate. It is a cationic bisbiguanide salt. CHG works by destroying the bacterial cell membrane and precipitating cell contents. The attraction of the positively charged CHG molecule to negatively charged bacterial cell wall causes disruption of the cell membrane and subsequent cellular death.1 CHG acts as a preservative to help inhibit microbial growth in the bottled solution.
What are Irrisept’s indications for use in the USA?
The Irrisept Antimicrobial Wound Lavage is intended for mechanical cleansing and removal of debris, dirt, and foreign materials, including microorganisms from wounds.
How much pressure is created through manual compression?
Irrisept is designed to meet the American College of Surgeons’ recommendations for low pressure irrigation (1-15psi).2
Where can I use Irrisept?
Irrisept can be used for all types of wounds.
Is Irrisept safe to use on wounds?
Irrisept features an extensive safety profile with in-vivo and in-vitro testing and RCT data.3,4 It has over 12 years of proven history and testing and has been referenced by name and/or concentration in independent publications.3-14
Irrisept meets biocompatibility guidelines for ≤ 24 hours contact with breached or compromised surfaces following the ISO standard 10993-1. The following testing supports biocompatibility of Irrisept for the intended use of wound cleansing and debridement3:
- Cytotoxicity
- Sensitization
- Irritation
- Acute Systemic Toxicity
- Material Mediated Pyrogens
Reports and testing on file at Irrimax Corporation.
What are the risks and safety information associated with use of Irrisept?
WARNINGS
- Do not use this product if the patient is allergic to chlorhexidine gluconate.
- Discontinue use immediately if irritation, sensitization, or allergic reaction occurs.
CAUTIONS
- Do not use unless solution is clear and bottle twist seal is intact.
- When using this product keep away from eyes and ear canals. If solution inadvertently contacts these areas, rinse out promptly and thoroughly with water and/or normal saline.
- Not for injection.
- Single patient use only.
- Irrisept is intended for use in adults by healthcare professionals only.
- Irrisept solution meets biocompatibility guidelines for ≤ 24 hours contact with breached or compromised surfaces (ISO 10993-1).
Why does Irrisept packaging state “Rx Only”?
Irrisept is a prescription device. FDA regulations require that prescription devices have the following statement “Caution: Federal law restricts this device to sale by or on the order of a licensed healthcare practitioner.”
Is Irrisept sterile?
Irrisept solution is manufactured using aseptic processing per ISO 13408 Aseptic Processing of Healthcare Products. The bottle, accessories, and packaging for the ISEPT-450-USA, ISEPT-150-USA, and ISEPT-AK-USA are EO sterilized.
Do I have to rinse with saline?
Labeling on all Irrisept products states: “Wait for approximately one minute, rinse with normal saline for irrigation. Discard any unused solution.”
Can Irrisept be used on more than one patient?
Labeling on all Irrisept products states: “Single patient use only” along with “single use only, do not reuse” symbol.
What are the storage condition requirements for Irrisept?
Labeling on all Irrisept products state storage temperature is between 10°–30° degrees Celsius (50°–86° Fahrenheit).
What is the shelf life of Irrisept?
Irrisept maintains a 3-year shelf life from the date of manufacture, which is indicated on the product packaging or IFU insert.
Where can I find the expiration date on the Irrisept product?
The expiration date may be located at the top of the Irrisept product insert or on a sticker on the pouch or box.
Can Irrisept be warmed?
If desired, the Irrisept Antimicrobial Wound Lavage may be warmed in its packaging up to 43°C (110°F) by placing it in a temperature warming cabinet for up to 15 days prior to use.15 Note: Prior to warming, store the device as per the device labeling.
What is the pH range for Irrisept?
Each LOT of Irrisept is tested to meet a pH range of 5.0 to 7.0.
References
1. McDonnell, G. & Russell, A.D. (1999). Antiseptics and Disinfectants; Activity, Action and Resistance. Clin Micro Rev, 12(1), 147-179. doi.org/10.1128/cmr.12.1.147
2. Ashley et al. (2014). Acute Wound Care. ACS Surgery: Princ Prac . (7th ed., pp. 215–216). Decker Intellectual Properties Inc.
3. Biocompatability Matrix. Data on file at Irrimax Corp. Lawrenceville, GA.
4. (2022). Protocol CLP-01: An Independent Review of Safety Data From a Closed Clinical Study Using Irrisept (Protocol # IRR-CT-901-2013-01)
5. Data on file at Irrimax Corp., Lawrenceville, GA
6. (2022). KTK Summary. Doc. 537161 V5 Evaluation of CHG as a preservative in the solution
7. Spencer et al. (2017). Reduction in Colon Surgical Site Infections Using CHG Irrigant Solution [Conference Presentation]. AORN 2017, Boston, MA
8. Truitt, K., & Kleinheinz, S. (2017). Target Zero: Eliminating Surgical Site Infection With 0.05% CHG Jet Lavage Irrigation [Conference Presentation]. AORN 2017, Boston, MA
9. Dotson, N., Rasheid, S., Marcet, J., & Sanchez, J. (2015). In Irrigation of Incisions With 0.05% CHG Reduces Surgical Site Infections in Colorectal Surgery [Conference Presentation]. ASCRS 2015, Boston, MA
10. Merceron et al. (2019). Comparison of Complications Following Implant-Based Breast Reconstruction Using Triple Antibiotic Solution Versus Low Concentration Chlorhexidine Gluconate Solution. Mod Plas Surg, 09(04), 74–85. https://doi.org/10.4236/mps.2019.94010
11. Mangold et al. (2019). Standardising Intraoperative Irrigation with 0.05% Chlorhexidine Gluconate in Caesarean Delivery to Reduce Surgical Site Infections: A Single Institution Experience. J Peri Practice, 30(1-2), 24–33. https://doi.org/10.1177/1750458919850727
12. Lung et al. (2022). Chlorhexidine Gluconate Lavage During Total Joint Arthroplasty May Improve Wound Healing Compared to Dilute Betadine. J Exp Ortho, 9(1). https://doi.org/10.1186/s40634-022-00503-w
13. Driesman et al. (2020). Perioperative Chlorhexidine Gluconate Wash During Joint Arthroplasty has Equivalent Periprosthetic Joint Infection Rates in Comparison to Betadine Wash. J Arthroplasty, 35(3), 845–848. https://doi.org/10.1016/j.arth.2019.10.009
14. Frisch et al. (2017). Intraoperative Chlorhexidine Irrigation to Prevent Infection in Total Hip and Knee Arthroplasty. Arth Today, 3(4), 294–297. https://doi.org/10.1016/j.artd.2017.03.005
15. TR 17-017 Irrisept Temperature Excursion Test